Know Your Iron - 2
In the last blog titled "Know Your Iron - 1", I gave you chapter and verse on this fascinating metal. You were also introduced to one-off company SGF fab. Precast handling crane manufacturers SGF Fab have made a name for themselves in the construction equipage industry. These goliath crane manufacturers provide infallible cranes, material handling equipment, and industrial fabrications that escalate a gamut of activities. What is a cherry on top? SGF Fab customize implements for different businesses as per needs. You have an a la carte menu and a set meal to choose from!
In today's blog, we will discuss Fe in detail. Have a gander at Fe's profile -
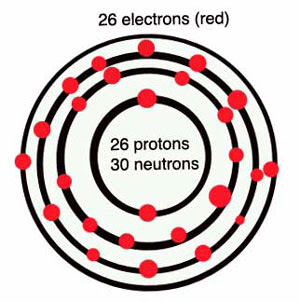
· Atomic number (number of protons in the nucleus): 26
· Atomic symbol (on the Periodic Table of Elements): Fe
· Atomic weight (average mass of the atom): 55.845
· Density: 7.874 grams per cubic centimeter
· Phase at room temperature: Solid
· Melting point: 2,800.4 degrees Fahrenheit (1,538 degrees Celsius)
· Boiling point: 5,181.8 F (2,861 C)
· Number of isotopes (atoms of the same element with a different number of neutrons): (includes how many are stable isotopes): 33 Stable isotopes: 4
· Most common isotopes: Iron-56 (natural abundance: 91.754 percent)
Don't fret if you are incognizant of the terms "atomic number" or "atomic symbol". These facts are just snippets of information.
Credits - quatr.us
Where does iron come from?
Iron is mostly obtained from minerals hematite and magnetite. In smaller degrees, it can also be obtained from the minerals taconite, limonite and siderite, according to Jefferson Lab. Iron has four different allotropic forms. Allotropy is the property of a chemical element to exist in two or more different forms in the same physical state. Here, it also means that Fe has four different structural forms in which atoms bond in different patterns, according to the Los Alamos National Laboratory. These forms are called ferrites, known as alpha (which is magnetic), beta, gamma, and omega.
Iron and Human Body
Iron is an important nutrient in our diet. These vegetarian foods are glutted with iron - lentils, soybeans, tofu, nuts and seeds, mushrooms, potato, et al. Iron deficiency, the most common nutritional deficiency, can cause anemia and fatigue that affects the ability to perform physical work in adults.
Lack of iron in the diet causes peevishness, dizziness, and headache. It can also impair memory and sundry mental functions in teens, according to the Centers for Disease Control and Prevention. In a nutshell, eat your vegetables and fruits!
The quantity of iron that is to be consumed by an adult varies according to a person's gender and life stage. For instance - Men and post-menopausal women generally require around 8 mg of iron per day. This amount increases to 18 mg per day for menstruating women and to 27 mg per day for pregnant women. Women who are iron deficient while pregnant are at an increased risk of having small and early babies, the CDC warns.
There are two types of dietary iron: heme iron and non-heme iron. Heme iron — which is the more readily absorbed type of iron — is found in meat, fish and poultry, whereas non-heme iron — which is also absorbed but to a lesser extent than heme iron — is found in both plant foods (such as spinach, kale and broccoli) and meat, according to the American Red Cross. People absorb up to 30 percent of heme iron, compared with 2 to 10 percent of non-heme iron, the ARC reports, adding that foods rich in vitamin C such as tomatoes or citrus fruits can help absorb non-heme iron.
(Inputs from live science)
___________________________________________________
Comments
Post a Comment